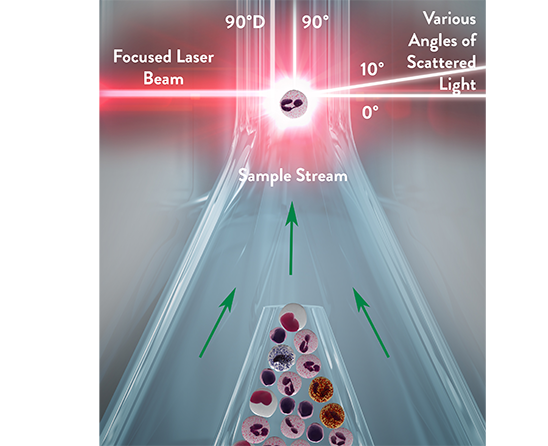
The Multi-Angle Polarized Scatter Separation (MAPSS™) technology from Abbott uses four light scatter detectors to determine various cellular features. The application of a depolarized light detector is a unique characteristic of this method and allows for specific identification of eosinophil granulocytes.
The four detectors generate the following signals:
- 0° or Axial Light Loss (ALL): related to size
- 0° to 10° Intermediate Angle Scatter (IAS): related to cellular complexity
- 90° Polarized Side Scatter (PSS): related to nuclear lobularity/segmentation
- 90° Depolarized Side Scatter (DSS): related to eosinophil granules
These signals correlate with morphological characteristics that can be determined visually under the microscope from a stained slide. Various combinations of these four measurements are used to classify the WBC subpopulations and provide morphological flagging. Fluorescent flow cytometry is used to detect NRBCs based on DNA staining when additional reagent and a detector for fluorescence signal is added.1
Multi-dimensional cell classification using patented Multi Angle Polarized Scatter Separation (MAPSS™) plus 3-color Fluorescent technology provides analysis of both the White Blood Cell count and the differential. MAPSS™ plus 3-color Fluorescence uses fluorescent flowcytometric measurements of multiple angles of light scatter of up to 20,000 cellular events. This unique process enables multiple scatterplots to be produced to further the characterization of cellular events.1
MAPSS™ technology provides:
- Identification and enumeration of WBCs and subpopulations
- Identification of abnormal cell types
- Identification of interfering substances thereby giving:
– First pass identification of NRBCs.
– Accurate Identification of Platelet Clump.1